Molar mass is measured in grams per mole. The mass in grams of one mole of a material is known as molar mass. For a molecule or compound, simply add up all the molar masses of the elements, taking subscripts into account. What is molar mass, and what is its unit? The molar mass of any element is on the periodic table. The mass of a specimen of a chemical compound divided by the amount of material in that specimen, expressed in moles, is the molar mass of that compound.ģ. So, in our example, carbon has a molar mass of 12.01 grams per mole. The molar mass is a bulk, not molecular, property of a substance. For example, if you want to find the molar mass of carbon, you would find the atomic mass of carbon on the periodic table, and this is equal to the molar mass in grams per mole. In chemistry, the molar mass (M) of a chemical compound is defined as the ratio between the mass and the amount of substance (measured in moles) of any sample of said compound. Add everything up and convert to grams/mole. The molar mass of elements is found by looking at the atomic mass of the element on the periodic table.Multiply every element’s atomic weight by the number of atoms included in the compound.Calculate the number of atoms of every element in the compound using the chemical formula.The standard unit is g mol⁻¹ and the SI one as kg mol⁻¹.Molar mass can be defined as the smallest unit of mass of any compound and is the twelfth of one carbon mass – 12 atoms.Find the number of moles in a 39.25-gram sample of CdBr₂.

Calculate the molar mass of KAl(SO₄)₂♱2 H₂O.Here are some questions for you to practise: Let’s take another example to find the mass of sulfur in an 8.5 mol sample of sulfur: To get the final answer, add up the total values, i.e., 46 12 48 = 106Hence, the molar mass of Na₂CO₃ is 106 g/mole. Now that you know the atomic mass and how many atoms of the compound are involved, you can find the molar mass as shown below. Count the number of atoms present in each element (The subscript of an element in its chemical formula).For any element, the number that appears on the top left represents atomic mass. For each element, you must figure out the atomic mass using the periodic table. 10.5 kg of Na 2 SO 4 ♱0H 2 O in 18.60 L of solution. 0.2074 g of calcium hydroxide, Ca (OH) 2, in 40.00 mL of solution. 98.0 g of phosphoric acid, H 3 PO 4, in 1.00 L of solution. Using the periodic table, find the atomic masses. Determine the molarity for each of the following solutions: 0.444 mol of CoCl 2 in 0.654 L of solution.Source HOW TO CALCULATE THE MOLAR MASS OF A COMPOUND? In any substance, the number of entities present can always be defined as a mole. The molar mass of any substance is defined as the smallest mass unit of that compound with one-twelfth of the mass of one carbon that would be 12 atoms. So, how do you estimate the molar mass mathematically? WHAT IS MOLAR MASS?

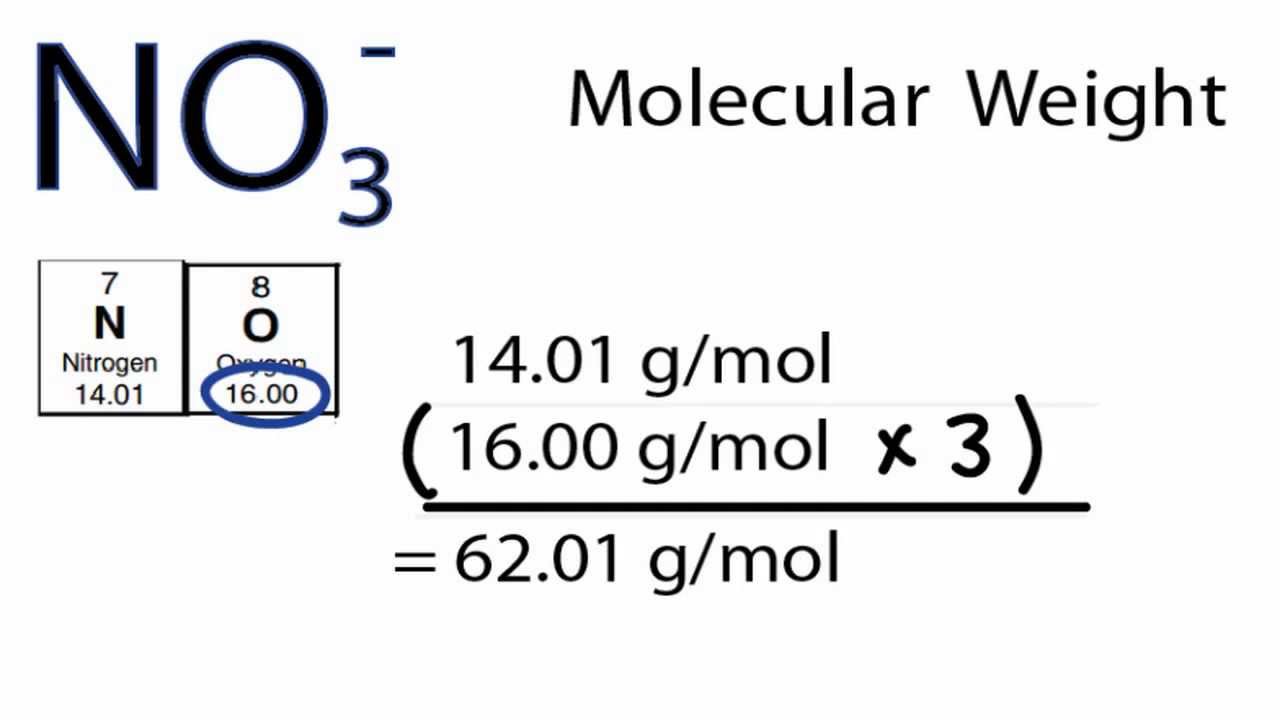
The molar mass of a compound is computed by summing the atomic masses of all the atoms in the molecule. The molar mass is the mass of one sampling mole. Where \(C\) and \(V\) are concentration and volume, respectively.Have you ever wondered how many air molecules there are in the soccer ball you’re playing with or the food you’re eating? Atoms or molecules are the smallest units in terms of mass and size, as we all understand. Reflecting this versatility, the dilution equation is often written in the more general form:
Although we derived this equation using molarity as the unit of concentration and liters as the unit of volume, other units of concentration and volume may be used, so long as the units properly cancel per the factor-label method. This relation is commonly referred to as the dilution equation. Thus, these two equations may be set equal to one another: Since the dilution process does not change the amount of solute in the solution, n 1 = n 2. Where the subscripts “1” and “2” refer to the solution before and after the dilution, respectively. According to the definition of molarity, the molar amount of solute in a solution is equal to the product of the solution’s molarity and its volume in liters:Įxpressions like these may be written for a solution before and after it is diluted: The liquid in the glass container is almost black in color.Ī simple mathematical relationship can be used to relate the volumes and concentrations of a solution before and after the dilution process. In the second photo the powder and about half the water have been added to the glass container. In the first, there is an empty glass container, 4.75 g of K M n O subscript 4 powder on a white circle, and a bottle of distilled water. (credit: modification of work by Mark Ott) This figure shows two photos. \): A solution of \(KMnO_4\) is prepared by mixing water with 4.74 g of KMnO4 in a flask.

